15
MAY 2023
Rescue Wrap – Emergency Transport Wrap
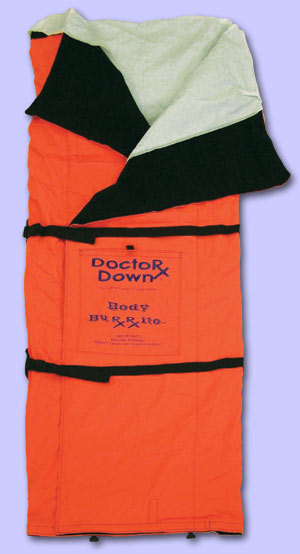
Posted by Doctor Down Rescue Wrap | Products, Rescue Wrap, Transport WrapThe Rescue Wrap Ranger is the best all-weather emergency wrap designed for medical transport.
The Rescue Wrap Ranger meets the specific needs of specialized trauma teams and offers maximum protection when wet weather or helicopter down-draft is a factor.
The black shell and carry handles make it desirable for military operations where quick-response and security issues are critical. The patented system addresses the
challenge of providing comfort, easy unlimited patient access and environmental protection for the patient and provider. It is durable, versatile and washable.
The fluid-proof, wind proof, breathable shell encases the synthetic down insulation cores. The cores velcro into place and can be added or removed to meet
the needs of the patient and conditions.
The disposable liners velcro in to place and can be replaced each time the Rescue Wrap Ranger is used so a new, clean surface is provided for every casualty.
Special features of the Rescue Wrap® Ranger
• Compact, lightweight and easy to carry ( approx. 12 lbs.)
• Trip report secures in chest pocket
• 360° patient access
• Straps secure IV, monitor & oxygen equipment to the Rescue Wrap® Ranger
• Can be modified for temperatures from 75° F to -40° F
• Quick Grip Handles used for easy movement of patient
• Accommodates virtually any size patient
• Overall dimensions of closed Rescue Wrap® Ranger: 36″ x 80″
Other Doctor Down® Products Available for the Rescue Wrap® Ranger :
• Attachable Hood (Fleece Lined)
• Reusable Heat/Cold Packs
• IV Warm Wrap
• Disposable Liners
• Husky Panels
NSN 6515-01-504-6091 Rescue Wrap Insulated Black
Loaded with 2 Primaloft® insulated cores, top & bottom shell, 1 set of Industrial Disposable Liners, one 8″x8″ Reusable Heat Pack, instruction manual, complete unit packed in Wrap Sack.
Dimensions: 37″ x 80″ (Girth 70″) 14 lbs. / 6.35 kg.
Call Doctor Down Inc in Polson Montana at : (406)883-3052